Introduction
Something is changing in your body and you know it - even if no one has adequately explained what, or why.
Maybe the weight is accumulating around your middle despite nothing obvious changing in your diet or activity. Maybe you are sleeping badly in a way that feels qualitatively different from before. Maybe the energy crashes, the brain fog, the mood swings, or the anxiety have become a persistent backdrop to daily life. Maybe your body is simply not responding the way it used to - to the food choices that previously worked, to the exercise that previously helped, to the approaches that previously felt adequate.
You are not imagining any of it. And you are not doing anything wrong.
What you are experiencing is a genuine metabolic transition - one that is driven by profound and systematic changes in the hormonal environment of your body, and that affects virtually every system simultaneously. It is one of the most significant biological transitions a woman goes through, and it is one of the least well-explained in mainstream healthcare.
This guide exists to change that.
What follows is a comprehensive, clinically grounded explanation of what perimenopause does to metabolism - why it happens, what the mechanisms are, what the consequences look like in daily life, and what an evidence-based approach to managing it actually involves. Not a list of tips. Not a reassurance that "this is normal." A genuine clinical framework that gives you the understanding to work with your body rather than against it in this chapter.
Start Here: Reset Your Metabolism
If you want a structured starting point, download our free 7-day metabolic reset guide.
A simple, clinically-informed plan to help stabilise blood sugar, improve metabolism, and reduce common hormonal symptoms.

What Is Perimenopause? Defining the Transition
Perimenopause is the hormonal transition that precedes menopause - defined as twelve consecutive months without a menstrual period. It is not a brief event. It is a sustained biological process that typically spans four to ten years, during which the ovaries progressively reduce their production of oestrogen and progesterone.
In practical terms, perimenopause most commonly begins in the early-to-mid 40s - though hormonal changes can begin as early as the late 30s. The average age of menopause in Western countries is 51–52, which means many women are in some phase of the perimenopausal transition for a significant portion of their 40s.
The hormonal pattern of perimenopause is not a simple, linear decline. It is characterised by fluctuation - oestrogen levels that rise and fall unpredictably, sometimes spiking higher than premenopausal levels before dropping sharply. Progesterone declines earlier and more steadily than oestrogen, creating a period of relative hormonal imbalance before both eventually reach their post-menopausal baseline.
This volatility - not just the eventual decline - is the source of much of the symptom burden in perimenopause. The body is not simply adjusting to lower hormones. It is adapting to a constantly shifting hormonal environment for which there is no stable baseline.
Perimenopause is not the beginning of the end of your hormonal life. It is a transition - a complex, demanding, and deeply consequential one - but a transition nonetheless. Women who understand what is happening, why their body is responding the way it is, and what evidence-based management looks like consistently report better outcomes and better quality of life through the transition than those who are managing without that framework. Understanding is the most powerful clinical tool available.
The Hormonal Changes of Perimenopause: What Is Actually Happening
To understand the metabolic consequences of perimenopause, you need to understand the hormonal changes that drive them.
Oestrogen: Decline, Fluctuation, and Loss of Metabolic Protection
Oestrogen - primarily oestradiol (E2) in the reproductive years - is far more than a reproductive hormone. It is a metabolic hormone with direct effects on insulin sensitivity, fat distribution, inflammatory regulation, bone metabolism, cardiovascular function, neurological health, and mood.
In perimenopause, oestradiol production by the ovaries becomes increasingly erratic and eventually declines to very low levels. This is not simply the loss of a reproductive signal. It is the withdrawal of a metabolic regulator that has been maintaining insulin sensitivity, managing fat storage patterns, modulating inflammatory responses, and protecting neurological function throughout the reproductive years.
The metabolic consequences of oestrogen withdrawal are substantial and systematic - and they are covered in detail throughout this guide.
Progesterone: The Early Decline
Progesterone declines before oestrogen in the perimenopausal transition - and its deficiency creates a specific and clinically significant hormonal imbalance.
As ovulation becomes less frequent, the corpus luteum - which produces the bulk of progesterone in the luteal phase - forms less reliably. Progesterone levels in the second half of the cycle drop, and eventually cycles become entirely progesterone-deficient as ovulation ceases.
Progesterone has direct effects beyond cycle regulation: it is the precursor to the neurosteroid allopregnanolone, a potent GABA-A receptor modulator that is the brain's primary natural anxiolytic. It supports sleep architecture, modulates cortisol, and opposes the proliferative effects of oestrogen on the uterine lining. Its progressive withdrawal has wide-ranging consequences - for mood, anxiety, sleep, and ultimately metabolic regulation.
Testosterone: Gradual Decline With Age
Testosterone declines gradually through the 40s - a process that begins earlier than most women expect and that is independent of the oestrogen and progesterone changes of perimenopause. The consequences include reduced libido, reduced energy, reduced muscle mass and strength, and a shift in body composition toward higher fat and lower lean mass.
For women with PCOS, whose testosterone was previously elevated, the decline toward more normal levels may produce some modest improvement in androgen-driven symptoms - though the net androgenic environment can worsen as oestrogen falls, because oestrogen had been partially offsetting the clinical expression of androgen activity.
FSH and LH: The Rising Signals
As ovarian function declines, the pituitary responds by increasing FSH (follicle-stimulating hormone) output - working harder to stimulate increasingly unresponsive ovaries. Rising FSH is the most accessible blood marker of the perimenopausal transition, though its utility is limited by significant cycle-to-cycle variability in early perimenopause.
LH also shifts - with implications for the signalling that governed the cycle throughout the reproductive years. The HPO (hypothalamic-pituitary-ovarian) axis that orchestrated ovulation and hormonal cycling gradually loses its coordinating function as ovarian responsiveness declines.
How Perimenopause Changes Metabolism: The Core Mechanisms
This is the central clinical section of this guide - and the one most likely to explain the changes you have been experiencing without adequate explanation.

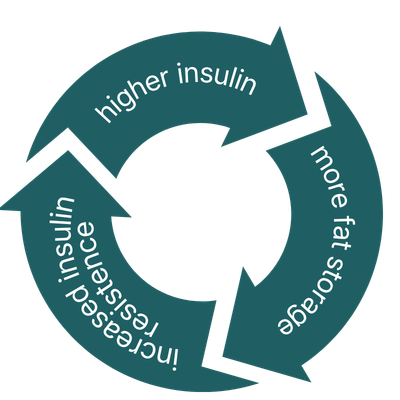
Insulin Resistance Worsens as Oestrogen Falls
Oestrogen has direct insulin-sensitising effects on muscle, fat, and liver tissue. It upregulates GLUT4 transporters - the mechanisms by which cells absorb glucose - enhances insulin receptor sensitivity, and supports mitochondrial efficiency in glucose metabolism.
As oestrogen declines in perimenopause, these insulin-sensitising effects are progressively withdrawn. Insulin resistance worsens - independently of diet, exercise, or any lifestyle factor - as a direct consequence of the hormonal transition.¹
The practical consequence is that the metabolic response to the same food choices changes. Carbohydrates that were previously well-managed now produce larger blood sugar spikes. Blood sugar instability becomes more pronounced. Fasting insulin rises gradually. And the risk of progressing toward type 2 diabetes increases - with studies showing that the years around menopause represent a clinically significant inflection point in glucose metabolism for many women.
For women who already have insulin resistance - from PCOS, from metabolic syndrome, or from long-standing dietary patterns - this oestrogen-withdrawal effect compounds an already compromised picture. As explored in PCOS After 40: When PCOS and Perimenopause Start to Overlap, the double-hit of PCOS-driven and oestrogen-withdrawal insulin resistance creates a metabolic challenge that requires a correspondingly more comprehensive approach.
1. Mauvais-Jarvis F, et al. (2013). The role of oestrogens in insulin resistance: a review. Trends in Endocrinology and Metabolism, 24(5), 251–258.

Fat Redistribution: Why the Abdomen Changes
One of the most consistently reported and most distressing metabolic changes in perimenopause is a shift in fat distribution - specifically, an increase in visceral and abdominal fat alongside relative loss of subcutaneous fat from the hips and thighs.
This is not simply weight gain. It is a genuine change in where and how the body stores fat - driven by the interaction of declining oestrogen, rising cortisol reactivity, and worsening insulin resistance.
Oestrogen promotes the subcutaneous (gluteofemoral) fat distribution pattern characteristic of premenopausal women. As oestrogen declines, this protective effect on fat distribution is withdrawn - and fat preferentially accumulates in visceral (abdominal) depots, which have a higher density of glucocorticoid and androgen receptors.
Visceral fat, as covered in PCOS and Inflammation, is hormonally and metabolically active tissue - producing inflammatory cytokines, contributing to insulin resistance, and amplifying androgen activity. The shift toward visceral adiposity in perimenopause therefore has metabolic consequences that go well beyond the cosmetic changes women observe.²
2. Lovejoy JC, et al. (2008). Increased visceral fat and decreased energy expenditure during the menopausal transition. International Journal of Obesity, 32(6), 949–958.

Muscle Mass Decline: The Metabolic Consequence of Sarcopenia
Oestrogen and progesterone both support muscle protein synthesis - they are anabolic hormones in the skeletal muscle context, supporting the maintenance of lean mass throughout the reproductive years.
As both hormones decline in perimenopause, muscle protein synthesis decreases, and the age-related loss of muscle mass - sarcopenia - accelerates. Women lose approximately 1–2% of muscle mass per year in the perimenopausal transition if no active countermeasures are taken.³
This matters metabolically because skeletal muscle is the primary site of insulin-mediated glucose disposal. Reducing muscle mass reduces the body's capacity to absorb and utilise glucose - directly worsening insulin resistance and contributing to the metabolic changes that accumulate through this transition.
Muscle loss also reduces resting metabolic rate - the number of calories the body burns at rest - which contributes to the weight gain many women experience in perimenopause even without changes in dietary intake.
Thyroid Function and the Perimenopausal Transition
Thyroid dysfunction becomes more prevalent in perimenopause - both because autoimmune thyroid disease (Hashimoto's) has its peak onset in the 40s and 50s for women, and because the hormonal changes of perimenopause directly affect thyroid hormone metabolism.
Declining oestrogen alters thyroid hormone binding, and the cortisol dysregulation common in perimenopause impairs T4-to-T3 conversion - reducing the availability of active thyroid hormone at the cellular level. The symptomatic overlap between perimenopause and hypothyroidism is substantial, and thyroid dysfunction is frequently missed in women whose symptoms are attributed entirely to the menopausal transition.
As detailed in PCOS and Thyroid, a full thyroid panel - including antibodies, free T3, and free T4, not just TSH - is a clinical priority for women in this transition who are experiencing significant fatigue, weight resistance, cognitive symptoms, or mood changes.

The HPA Axis in Perimenopause: Cortisol and Stress Reactivity
Oestrogen has a modulating effect on the HPA axis - it buffers cortisol reactivity and supports the return to baseline after stress. As oestrogen declines, this buffer is removed, and the HPA axis becomes more reactive. Cortisol responses to stressors become larger and last longer.
The metabolic consequences of this increased cortisol reactivity are direct and far-reaching - elevated cortisol drives gluconeogenesis (raising blood glucose), promotes visceral fat accumulation, suppresses thyroid hormone conversion, fragments sleep, and amplifies the inflammatory environment. All of these effects compound the insulin resistance and body composition changes already driven by oestrogen withdrawal.
For women who already have HPA axis dysregulation - from PCOS, from chronic life stress, or from years of inadequate sleep - the removal of oestrogen's buffering effect in perimenopause can produce a significant worsening of cortisol-related symptoms.
Full detail on the cortisol mechanisms: Cortisol and PCOS: The Stress–Blood Sugar Connection - the mechanisms described there apply with equal force, and additional urgency, in the perimenopausal context.

Neurological Changes: Brain Fog, Mood, and the Oestrogen–Brain Connection
Oestrogen has direct neuroprotective and neurotrophic effects - it supports synaptic plasticity, promotes the production of serotonin and dopamine, reduces neuroinflammation, and maintains the integrity of the blood-brain barrier.
The cognitive symptoms of perimenopause - word-finding difficulties, memory lapses, difficulty concentrating, the sensation of "brain fog" - are a direct neurological consequence of oestrogen withdrawal, not simply the psychological effect of poor sleep or stress. Research using neuroimaging shows measurable changes in brain metabolism and function during the perimenopausal transition that correlate with oestrogen levels.⁴
These cognitive changes are temporary in most women - brain function largely restores in the post-menopausal years as the brain adapts to the new hormonal environment. But during the transition, particularly in the context of insulin resistance (which independently impairs cognitive function), the combination can be significantly disruptive.
The serotonin and dopamine effects of declining oestrogen contribute directly to the mood changes, anxiety, and depression that are prevalent in perimenopause - operating through the same neurochemical pathways affected by the gut microbiome disruptions and inflammatory signalling discussed in PCOS and Gut Health and PCOS and Anxiety.
4. Mosconi L, et al. (2021). Menopause impacts human brain structure, connectivity, energy metabolism, and amyloid-beta deposition. Scientific Reports, 11(1), 10867.
Recognising the Symptoms of Perimenopausal Metabolic Disruption
The metabolic changes of perimenopause produce a recognisable symptom cluster. Understanding which symptoms are metabolically driven - rather than simply hormonal or psychological - changes what you do about them.
Vasomotor symptoms (hot flushes and night sweats) are the hallmark perimenopausal symptom - and they have a metabolic dimension that is rarely discussed. Hot flush frequency and severity are significantly worse in women with insulin resistance, higher BMI, and higher inflammatory load.⁵ This is not coincidental - it reflects the interaction between the thermoregulatory effects of oestrogen withdrawal and the metabolic environment in which that withdrawal occurs. Improving insulin sensitivity and reducing inflammatory load genuinely reduces vasomotor symptom burden.
Sleep disruption operates through multiple simultaneous mechanisms in perimenopause: night sweats causing waking, the direct sleep-architecture effects of progesterone withdrawal, increased cortisol reactivity disrupting sleep initiation and maintenance, and the rising FSH levels that have been shown to independently fragment sleep. As detailed in PCOS and Sleep, sleep disruption in this context is both a symptom and a metabolic driver - poor sleep worsens insulin resistance and cortisol reactivity, which worsen the perimenopausal metabolic picture further.
Weight gain and body composition changes - particularly the shift toward abdominal weight - are among the most universally reported and most distressing symptoms of perimenopause. They are metabolically driven, not simply caloric, and they respond to metabolic intervention rather than simple caloric restriction.
Fatigue in perimenopause has multiple metabolic contributors: worsening insulin resistance reducing cellular energy efficiency, declining thyroid conversion reducing metabolic rate, poor sleep compounding HPA axis dysregulation, and the direct energetic cost of hormonal volatility on the nervous system.
Mood changes, anxiety, and depression - driven by declining progesterone and its GABA-modulating effects, oestrogen withdrawal affecting serotonin and dopamine metabolism, neuroinflammation, and the psychological burden of significant physiological change.
Joint aches and pains - oestrogen has anti-inflammatory effects on joint tissue. Its withdrawal increases joint inflammation, producing the musculoskeletal symptoms that many women in perimenopause experience but rarely connect to the hormonal transition.
Cognitive symptoms - brain fog, memory changes, word-finding difficulties - driven by the neurological effects of oestrogen withdrawal compounded by insulin resistance, sleep disruption, and neuroinflammation.
Keeping a symptom diary through the perimenopausal transition - noting the timing, severity, and pattern of symptoms alongside sleep, stress, diet, and cycle data - serves two purposes. First, it provides clinically useful information for any practitioner you work with. Second, and perhaps more importantly, it helps you identify the metabolic and lifestyle factors that modulate your symptoms - giving you genuine agency over a transition that can otherwise feel entirely out of your control.
5. Thurston RC, et al. (2017). Vasomotor symptoms and insulin resistance in the Study of Women's Health Across the Nation. Journal of Clinical Endocrinology & Metabolism, 97(10), 3487–3494.
The Long-Term Metabolic Stakes
Understanding the long-term consequences of perimenopausal metabolic changes provides important context for why active management in this decade matters beyond symptom relief.
Cardiovascular Risk
Premenopausal women have significantly lower cardiovascular disease rates than men of equivalent age - a difference largely attributable to oestrogen's cardioprotective effects on vascular elasticity, inflammatory markers, and lipid profiles. The removal of this protection at menopause is the primary reason cardiovascular disease rates in women rise sharply in the post-menopausal years and eventually equalise with men.
The quality of the metabolic transition - how well insulin resistance, inflammatory load, blood pressure, and body composition are managed through perimenopause - directly determines the cardiovascular risk profile carried into menopause and beyond. This is not a distant, theoretical concern. It is a concrete clinical reason why metabolic management in the 40s has long-term cardiovascular relevance.
Type 2 Diabetes Risk
The progressive worsening of insulin resistance through perimenopause creates a clinically significant inflection point for type 2 diabetes risk. Women who enter perimenopause with pre-existing insulin resistance - from PCOS, metabolic syndrome, or long-standing dietary patterns - are at meaningfully elevated risk of progressing to impaired glucose tolerance or type 2 diabetes in the perimenopausal and early post-menopausal years. Active insulin resistance management during this transition is directly protective - and the dietary and lifestyle strategies covered throughout this content ecosystem are the primary clinical tools for reducing that risk.
Bone Health
Oestrogen is the primary regulator of bone density in women. Its decline in perimenopause initiates the most rapid phase of bone loss in a woman's lifetime - with some studies showing 10–15% of lifetime bone density lost in the first five years following menopause.
Bone health in the perimenopausal years is supported by adequate calcium and vitamin D, resistance training (mechanical loading stimulates bone remodelling), and - for women with significant risk factors or early bone density loss - clinical management including HRT.
Cognitive Health
The oestrogen withdrawal of menopause has long-term neurological consequences. The years around the menopausal transition represent a window of increased neurological vulnerability - and there is accumulating evidence that the quality of metabolic management during this period influences long-term cognitive health outcomes, including dementia risk, through mechanisms involving insulin resistance, neuroinflammation, and vascular health.⁷ This is an emerging area of research rather than established clinical guidance - but the direction of evidence consistently supports the same metabolic health priorities relevant to symptom management now.
7. Brinton RD, et al. (2015). Perimenopause as a neurological transition state. Nature Reviews Endocrinology, 11(7), 393–405.
If you’re unsure where to start…
The Metabolic Reset Guide walks you through exactly how to structure your meals and stabilise your metabolism in a simple, practical way.
What Evidence-Based Management Looks Like
Managing perimenopause well - from a metabolic perspective - requires addressing the mechanisms, not just the symptoms. Here is the clinical framework.
Make Blood Sugar Management Central
Given that insulin resistance worsens as a direct consequence of oestrogen withdrawal, and that this worsened insulin resistance drives or amplifies most of the major metabolic changes of perimenopause, blood sugar management is the highest-leverage clinical priority in this transition.
The dietary approach that most directly addresses this: build every meal around adequate protein and healthy fat, choose lower-glycaemic carbohydrates, avoid eating carbohydrates alone, and eat at regular intervals to prevent the blood sugar drops that drive cortisol and adrenal responses.
The precision required increases as the transition progresses - the approaches that managed blood sugar adequately at 38 may need to become more consistent and more targeted at 45. This is not failure. It is an appropriate response to a changed hormonal environment.
For the evidence-based dietary framework that underpins this approach, the resources developed for insulin resistance in PCOS apply with full force in the perimenopausal context: Best Diet for PCOS and Insulin Resistance and How to Balance Blood Sugar with PCOS
Prioritise and Protect Muscle Mass
Resistance training is the single most important exercise modality in the perimenopausal transition - addressing insulin resistance, protecting muscle mass, supporting bone density, improving body composition, and reducing inflammatory markers simultaneously.
The evidence is consistent: two to four resistance training sessions per week, with progressive overload over time, produces the most significant and durable metabolic benefits available through exercise in this phase. Adequate dietary protein - 1.6–2.0g per kilogram of body weight daily - is essential to support muscle protein synthesis alongside training.
Daily walking and moderate movement complement resistance training without the cortisol burden of high-intensity cardio that is counterproductive when HPA axis reactivity is already elevated.
Address Sleep as a Metabolic Priority
Sleep disruption in perimenopause is both a symptom and a metabolic driver - and addressing it actively, rather than accepting it as an inevitable feature of the transition, produces metabolic benefits that extend well beyond subjective wellbeing.
Practical sleep strategies including circadian rhythm anchoring, evening cortisol reduction, blood sugar management to prevent nocturnal hypoglycaemia, and optimal sleep environment all remain relevant - and in some women need to be supplemented with clinical management of vasomotor symptoms that are the primary cause of nocturnal waking.
Full detail: PCOS and Sleep
Reduce Inflammatory Load Deliberately
Chronic low-grade inflammation worsens in perimenopause - driven by the withdrawal of oestrogen's anti-inflammatory effects, increasing visceral adiposity, and rising cortisol reactivity. An anti-inflammatory dietary pattern - prioritising omega-3 fatty acids, polyphenol-rich plant foods, and diverse dietary fibre - is both protective and symptom-modifying.
As covered in PCOS and Inflammation, reducing inflammatory load has measurable downstream effects on insulin sensitivity, mood, sleep quality, and vasomotor symptom burden - making it one of the most leveraged single interventions available.
Consider Targeted Supplementation
Several supplements have specific evidence for supporting metabolic health and symptom management in perimenopause:
Magnesium glycinate - supports insulin sensitivity, reduces cortisol reactivity, improves sleep quality, and has evidence for reducing hot flush frequency and severity. One of the most broadly relevant supplements for this transition.
Omega-3 fatty acids (EPA + DHA daily) - directly anti-inflammatory, support cardiovascular health, and have evidence for improving mood and reducing vasomotor symptoms.
Vitamin D (dosed to correct deficiency confirmed by testing) - essential for insulin receptor function, immune regulation, bone health, and mood. Deficiency is prevalent and clinically significant.
Creatine monohydrate - emerging evidence specifically for perimenopause supporting muscle mass maintenance, cognitive function, and bone density. The evidence base is strengthening rapidly and warrants attention.
Inositol - where insulin resistance is a prominent feature, myo-inositol continues to be relevant through the perimenopausal transition, supporting insulin sensitivity through the same mechanisms documented in PCOS.
Ashwagandha (KSM-66) - for HPA axis support, cortisol reduction, and the anxiety and sleep disruption driven by increased stress reactivity in perimenopause.
Have the HRT Conversation
Hormone replacement therapy - particularly body-identical oestradiol and micronised progesterone - addresses the hormonal root of perimenopausal metabolic disruption directly, in ways that dietary and lifestyle intervention alone cannot fully replicate.
Body-identical HRT:
- Restores oestrogen's insulin-sensitising effects on muscle and fat tissue
- Reduces the rate of visceral fat accumulation
- Supports muscle mass maintenance alongside resistance training
- Reduces vasomotor symptoms, which indirectly improves sleep and cortisol regulation
- Provides cardiovascular protection when initiated in the perimenopausal window (the "timing hypothesis" - early initiation is cardioprotective, late initiation is not)
- Restores progesterone's GABA-modulating and sleep-supporting effects
The current evidence strongly supports HRT as a safe and effective option for the majority of women in perimenopause - the Women's Health Initiative concerns that led to widespread HRT discontinuation in the early 2000s applied to synthetic, non-body-identical formulations administered to older post-menopausal women, and have been substantially revised in the twenty years since.⁸
HRT is not appropriate for every woman - individual history, risk profile, and clinical context all matter. But it is a conversation that deserves to happen with a clinician who is genuinely current on the evidence, and who understands both the perimenopausal transition and the metabolic picture that precedes it.
Dietary and lifestyle intervention and HRT are not competing alternatives in perimenopausal management. They address different layers of the same clinical picture and work best in combination. HRT addresses the hormonal root of metabolic disruption. Dietary and lifestyle intervention optimises the metabolic environment in which those hormones operate. Women who use both tend to have better outcomes than those who use either in isolation - and neither is a substitute for the other.
8. Manson JE, et al. (2017). Menopausal hormone therapy and long-term all-cause and cause-specific mortality. JAMA, 318(10), 927–938.

Can You Improve Perimenopause by Fixing Your Metabolism?
This is the question that matters most, and the answer is genuinely encouraging.
You cannot stop the perimenopausal transition. The decline in ovarian function is a biological process that no dietary or lifestyle intervention reverses. But the experience of that transition - the severity of symptoms, the degree of metabolic disruption, the long-term health outcomes - is profoundly influenced by the metabolic environment in which it occurs.
What the research consistently shows is that women who enter perimenopause with better metabolic health - lower insulin resistance, lower inflammatory load, better body composition, more regulated cortisol - move through the transition with significantly less symptom burden than those whose metabolic health is compromised. And for women who begin addressing their metabolic health during the transition, meaningful improvements in symptoms and metabolic markers are achievable at any point.
In clinical practice, addressing metabolic health through perimenopause is associated with:
Reduced vasomotor symptoms - hot flush frequency and severity are measurably worse in women with higher insulin resistance and inflammatory load. Improving both produces real reductions in vasomotor burden, not just general wellbeing.
Improved body composition - addressing insulin resistance directly counters the hormonal drive toward visceral fat accumulation, producing changes in abdominal weight and body composition that caloric restriction alone cannot achieve.
Better sleep quality - stabilising blood sugar reduces nocturnal glucose instability and cortisol surges that cause night waking; reducing inflammatory load improves sleep architecture; managing cortisol helps restore the evening decline that allows sleep initiation.
More stable mood and reduced anxiety - the blood sugar stability, reduced neuroinflammation, and HPA axis regulation produced by metabolic intervention have direct neurological consequences that women consistently report as among the most meaningful improvements they experience.
Clearer cognition - insulin resistance independently impairs brain glucose metabolism and cognitive function. Improving insulin sensitivity produces measurable improvements in mental clarity and concentration that are not simply the secondary effect of better sleep.
Long-term cardiovascular and metabolic protection - the quality of metabolic management through perimenopause directly shapes the cardiovascular risk profile carried into the post-menopausal years. This is not a distant theoretical benefit. It is a concrete clinical reason why the effort invested now pays forward significantly.
It is important to be clear: metabolic intervention is not a complete substitute for clinical management of perimenopausal symptoms where that is warranted. For many women, HRT alongside a strong metabolic foundation produces the best outcomes - each addressing what the other cannot. But for women who are not yet at the point of needing pharmaceutical management, or who want to optimise the environment in which any hormonal treatment operates, metabolic intervention is the most evidence-aligned and durable approach available.
The transition is not optional. How you move through it is.
Perimenopause is not simply something that happens to you. It is a transition you actively navigate — and the metabolic choices made in this decade have a disproportionate influence on how the transition feels now and on the health you carry into the decades that follow. Women who approach this chapter with a coherent metabolic framework consistently report better outcomes than those who manage symptoms reactively as they arise. Understanding what is happening is the essential first step. Acting on that understanding is what changes the trajectory.
The Signs Your Metabolic Approach Is Working
Because the goal in perimenopause is metabolic and hormonal rather than purely symptomatic, the markers of meaningful progress extend well beyond any single measure. The scale, in particular, is an unreliable indicator of metabolic improvement in this transition - body composition changes, hormonal shifts, and inflammatory reductions often precede visible weight change by weeks or months.
Look instead for this broader picture of improvement:
Energy becomes more consistent - fewer afternoon crashes, less reliance on caffeine to maintain function, a more even energy distribution across the day. This is one of the earliest signs that blood sugar stability is improving.
Sleep quality gradually improves - falling asleep more easily, fewer night wakings, waking with a greater sense of restoration even before vasomotor symptoms fully resolve. Sleep improvement is both a sign of metabolic progress and an accelerator of it.
Hot flush frequency or severity reduces - this takes longer than energy and sleep improvements, typically two to three months, but is a meaningful clinical marker of reduced inflammatory load and improving insulin sensitivity.
Mood becomes more stable - less emotional volatility, reduced background anxiety, greater resilience to daily stressors. Women often describe this as feeling more like themselves again - a shift that reflects genuine neurological improvement rather than simply feeling better subjectively.
Cognitive clarity returns - word-finding becomes easier, concentration improves, the fog lifts progressively. This typically correlates with improving insulin sensitivity and reducing neuroinflammation.
Body composition shifts before weight does - clothes fitting differently around the waist and abdomen, improved muscle tone from resistance training, a general change in how the body feels and moves - often visible months before the scale reflects it.
Cravings reduce and appetite regulates - the urgency around sugar and carbohydrates diminishes as blood sugar stability improves and insulin demand decreases. This shift is both a sign of progress and a reinforcing positive cycle - reduced cravings make dietary management easier, which further improves metabolic function.
Measurable improvements on retesting - fasting insulin falling toward the optimal range, hs-CRP reducing, triglycerides improving, SHBG rising. These objective markers tell you what the symptomatic picture cannot: that the internal metabolic environment is genuinely shifting.
The timeline for perimenopausal metabolic improvement is realistic but requires patience. Meaningful symptomatic change typically becomes apparent over two to four months of consistent effort - with laboratory markers improving over three to six months. This is the timescale of hormonal recalibration, not the timescale of a short-term diet.
Progress is cumulative. Each consistent day builds on the last. And the women who navigate this transition best are consistently those who commit to the metabolic foundation - not perfectly, but persistently.
Retest your core metabolic markers - fasting insulin, hs-CRP, fasting glucose, lipid panel, vitamin D, and thyroid panel - at three to six months after beginning a targeted metabolic approach. Tracking objective data alongside subjective symptoms gives you a complete picture of progress that motivation and willpower alone cannot provide. It also tells you whether your current approach needs adjustment - and where. Numbers are not the whole story, but they are a crucial part of it.
Where to Go From Here: The Perimenopause Content Ecosystem
This guide provides the clinical framework for understanding perimenopause and metabolism. The articles that follow in this content ecosystem go deeper on each component - explaining the specific mechanisms, the evidence, and the practical strategies in detail.
Understanding the transition:
Core metabolic mechanisms:
- Perimenopause and Insulin Resistance: Why Blood Sugar Changes in Your 40s
- Perimenopause and Cortisol: Why Stress Hits Harder in Midlife (coming soon)
- Perimenopause and Inflammation: The Metabolic Amplifier (coming soon)
Symptoms and specific concerns:
- Perimenopause and Weight Gain: What's Actually Happening
- Perimenopause and Sleep: Why You Can't Sleep and What Helps
- Perimenopause and Anxiety: The Hormonal Root Cause (coming soon)
- Perimenopause and Brain Fog: The Cognitive Changes Explained (coming soon)
Solutions:
- The Best Diet for Perimenopause and Metabolic Health (coming soon)
- Perimenopause and Exercise: What Works and Why (coming soon)
- Perimenopause Supplements: What the Evidence Says (coming soon)
Clinical Insight
The metabolic changes of perimenopause are among the most significant and systematic biological transitions in a woman's lifetime - and they are among the most inadequately addressed in mainstream healthcare.
The mechanisms are well-characterised in the clinical research:
- oestrogen withdrawal directly worsens insulin sensitivity,
- shifts fat distribution toward visceral adiposity,
- accelerates muscle loss,
- increases HPA axis reactivity, and
- removes cardioprotective and neuroprotective effects that were maintained throughout the reproductive years.
These are not incidental or subjective changes. They are measurable, mechanistically understood metabolic events with real consequences for long-term health. Managing them well - through evidence-based dietary intervention, targeted exercise, appropriate supplementation, and clinical management where indicated - produces measurably better metabolic outcomes both during the transition and in the post-menopausal years.
A woman who navigates this transition with clinical support and a coherent metabolic framework is not just managing symptoms. She is actively shaping her long-term health trajectory.
When to Seek Clinical Support
Perimenopause is not simply "hormones changing." It is a fundamental metabolic transition - one that alters insulin sensitivity, fat distribution, muscle mass, inflammatory load, cortisol reactivity, neurological function, and cardiovascular risk simultaneously.
Understanding that changes how you approach it.
The symptoms you are experiencing are not imagined, not inevitable, and not simply something to be endured. They are the output of a specific hormonal and metabolic mechanism - and they are responsive to a specific, evidence-based clinical approach.
That approach starts with the metabolic foundation: blood sugar management, anti-inflammatory nutrition, muscle-preserving exercise, sleep as a clinical priority, and cortisol regulation. It is complemented by targeted supplementation where evidence supports it and by HRT where clinically appropriate. And it is most effective when it is personalised - built around your individual metabolic picture, your specific symptoms, and where you are in the transition.
This is manageable. With the right framework and the right support, this transition can be navigated with your metabolic health intact - and in some respects, improved - on the other side.
Managing Your Metabolism Through Perimenopause?
In my clinical practice, I specialise in the clinical assessment and management of metabolic health in women through perimenopause and beyond - understanding the compound effects of hormonal change on insulin sensitivity, inflammation, body composition, and long-term health.
Our Metabolic Balance® programme uses your individual blood chemistry to design a personalised nutrition protocol calibrated to your current hormonal and metabolic environment - not a generic perimenopause diet, but a programme built around exactly where your body is right now.
For women navigating the perimenopausal metabolic transition, a whole-system, personalised approach is the most clinically complete and durable path forward.
In-clinic and remote consultations available. This transition deserves proper clinical support - we are here to provide it.
Start Improving Your Metabolic Health Today
Not ready for a full programme yet? Start with the reset.
The 7-Day Metabolic Reset is a free, structured guide for women navigating metabolic changes through perimenopause and insulin resistance - covering blood sugar stabilisation, anti-inflammatory nutrition, and practical daily strategies in a clear seven-day framework.
Inside, you will find:
- How to structure your meals for stable blood sugar and reduced insulin demand
- The key dietary drivers of symptoms - and how to address them
- Practical steps you can begin immediately, without overhauling everything at once
Clinically grounded. No gimmicks. A genuine starting point for working with your biology rather than against it.
